Drug name
Last update: Apr 2026Risperidone
Developer(s)
Drug information
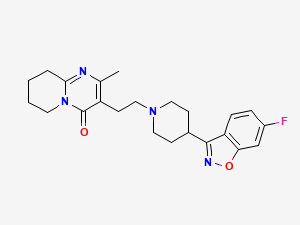
Risperidone
RISPERDAL CONSTA®, PERSERIS®, RYKINDO®, UZEDY®
Small molecule
Not provided
Risperidone LAI is an atypical antipsychotic indicated for schizophrenia and maintenance treatment of Bipolar I Disorder. It acts via dopamine D₂ and serotonin 5-HT₂ receptor antagonism. The apparent half-life for risperidone + hydroxyrisperidone is 3–6 days, and clearance is 13.7 L/h (in extensive metabolisers) and 3.3 L/h (in poor metabolisers). Following intramuscular injection, risperidone is released gradually, with therapeutic levels sustained for up to 6 weeks. It is metabolised hepatically via CYP2D6 to active 9-hydroxyrisperidone. Common adverse effects include extrapyramidal symptoms, weight gain, and hyperprolactinemia. Clinical trials demonstrate efficacy in reducing relapse rates in schizophrenia and bipolar disorder, with a favourable safety profile under monitored use.
Risperidone microsphere LAI formulation is approved in 54 countries including Canada, United States, European Union countries, UAE, Switzerland, Brazil, Chile, China, Russia, Hong Kong, Malaysia, India, New Zealand, Australia, Israel, Philippines, Saudi Arabia, South Africa, Taiwan, Thailand, the United Kingdom, South Korea, Turkey, Japan, United Arab Emirates, Argentina and Mexico.
Risperdal Consta (12.5 mg, 25 mg, 37.5 mg, and 50 mg) has received marketing authorization from multiple regulatory agencies, including Health Canada, the U.S. FDA, and authorities such as DOH, Swissmedic, ANVISA, ISP, NMPA, MINZDRAV, NPRA, CDSCO, MedSafe, TGA, MOH, FDA Philippines, SFDA, SAHPRA, TFDA, THAI FDA, MHRA, MFDS, TMMDA, PMDA, MOHAP, ANMAT, and COFEPRIS. In addition, approvals have been granted by European national agencies such as CBG, AGES, FAMHP, ANMDMR, SUKL (Czechia), SUKL (Slovakia), DMA, IMA, FIMEA, ANSM, BFARM, EOF, NNGYK, and HPRA. Other European authorities have also provided authorization. However, the European Medicines Agency (EMA) has issued only a positive opinion following a referral from BFARM (Germany), rather than granting centralized approval.
Therapeutic area(s)
- Mental health : "incl. schizophrenia"
- Treatment
Administration route
Oral, Subcutaneous, Intramuscular
Associated long-acting platforms
Polymer-based particles, Microspheres
Use of drug
- Administered by a nurse
- Administered by a specialty health worker
- Administered by a community health worker
- Every 2 weeks
Not provided
Dosage
12.5 mg; 25 mg; 37.5 mg; 50 mg powder for injection
50 mg IM every 2 weeks
1. Initial Treatment Dose for Schizophrenia & Bipolar: • Administer 25 mg intramuscularly (IM) every 2 weeks. 2. Dose Adjustment for Non-Responders: • If inadequate response at 25 mg, consider increasing to 37.5 mg or 50 mg IM every 2 weeks. • Do not exceed 50 mg every 2 weeks. 3. Special Populations (Renal or Hepatic Impairment): • Initiate treatment at a reduced dose of 12.5 mg IM every 2 weeks. 4. Previously Treated Patients: • Previous antipsychotics should be continued for 3 weeks after the first injection of risperidone LAI to ensure that therapeutic concentrations are maintained until the main release phase of risperidone from the injection site has begun
Risperidone LAI must be reconstituted only in the diluent supplied in the dose pack and must be administered with only the appropriate needle supplied in the dose pack for gluteal (2-inch needle) or deltoid (1-inch needle) administration. All components are required for administration. Do not substitute any components of the dose pack. To ensure that the intended dose of risperidone is delivered, the full contents from the vial must be administered. Administration of partial contents may not deliver the intended dose of risperidone.
Comment & Information
Developer(s)

Janssen/Johnson & Johnson
Janssen Pharmaceuticals is a subsidiary company of Johnson & Johnson headquartered in Beerse, Belgium. They focus on manufacturing and developing pharmaceutical products for use in areas such as, Immunology, Infectious Diseases & Vaccines, Pulmonary Hypertension, Cardiovascular & Metabolism, Oncology, and Neuroscience.

Alkermes Inc
Alkermes, Inc. is the U.S. R&D and manufacturing arm of Alkermes plc, a fully integrated, global biopharmaceutical company. The company focuses on developing innovative medicines for serious psychiatric and neurological disorders.
Drug structure
Scale-up and manufacturing prospects
Not provided
Static Mixer: Cole Parmer L04667-14. Gear Pumps: Cole Parmer L07149-04, L07002-16. Quench Tank: 55 L capacity. Sieves: 25 µm and 180 µm stainless steel mesh. Lyophilizer: Pilot-scale, ramped cycle (max 30°C, 50 h). Gamma Irradiator: Co-60 source.
Risperidone microspheres are synthesized by dissolving PLGA (75:25 lactide:glycolide) in ethyl acetate and benzyl alcohol, then adding risperidone base. This organic phase is emulsified with an aqueous phase containing 1% polyvinyl alcohol using a static mixer. The emulsion is quenched in chilled water with buffer, stirred for solvent removal, sieved (25–180 µm), washed, and dried. Optional steps include lyophilization and gamma sterilization for stability and sterility, ensuring controlled release for 14–60 days.
1. Laser Diffraction Analyzer 2. Scanning Electron Microscope (SEM) 3. High-Performance Liquid Chromatography (HPLC) 4. UV-Vis Spectrophotometer 5. Gas Chromatography (GC) 6. Differential Scanning Calorimetry (DSC) 7. Thermogravimetric Analysis (TGA)
Excipients
Not provided
The list of excipients used is: 1. Polysorbate 20 2. Sodium carboxymethyl cellulose 3. Disodium hydrogen phosphate dihydrate 4. Citric acid anhydrous 5. Sodium chloride 6. Sodium hydroxide 7. Water for injection
Not provided
Delivery device(s)
No delivery device
Microparticles
The invention provides a process for the preparation of biodegradable biocompatible microparticles, said process comprising: contacting microparticles comprising a biodegradable biocompatible polymer matrix containing an active (e.g. pharmaceutical or diagnostic) agent and an organic solvent with an aqueous solvent system whereby the content of said organic solvent in said particles is reduced to 2 % or less of the weight of said particles, said solvent system being such as to satisfy at least one of the conditions (a) that it is at an elevated temperature (e.g. from 25 to 40 °C) during at least part of the time that it is in contact with said particles, and (b) that it comprises water and water-miscible solvent for said organic solvent; and recovering said particles from said aqueous solv
WO1997041837A2
Not provided
Alkermes Controlled Therapeutics Inc
Not provided
Not provided
Not provided
Microencapsulated 3-piperidinyl-substituted 1,2-benzisoxazoles and 1,2-benzisothiazoles
Not provided
WO1995013814A1
Not provided
Janssen Pharmaceutica N.V.
Not provided
Not provided
Not provided
Publications
Eerdekens, Mariëlle et al. “Pharmacokinetics and tolerability of long-acting risperidone in schizophrenia.” Schizophrenia research vol. 70,1 (2004): 91-100. doi:10.1016/j.schres.2003.11.001
The pharmacokinetics and tolerability of long-acting risperidone (Risperdal Consta) were evaluated in a multicenter, prospective, open-label, 15-week study of 86 patients with schizophrenia. Subjects stabilized on 2, 4 or 6 mg of oral risperidone once daily for at least 4 weeks were assigned to receive i.m. injections of 25, 50 or 75 mg of risperidone, respectively, every 2 weeks for 10 weeks. The 90% confidence intervals for the i.m./oral ratios of the mean steady-state plasma-AUC, corrected for dosing interval, and of the average plasma concentration of the active moiety (risperidone plus 9-hydroxyrisperidone) were within the range of 80-125%, indicating bioequivalence of the i.m. and oral formulations. However, mean steady-state peak concentrations of the active moiety were 25-32% lower with i.m. than oral dosing (P < 0.05) and fluctuations in plasma active-moiety levels were 32-42% lower with the i.m. than oral regimen. Symptoms of schizophrenia continued to improve after switching from oral to i.m. dosing. Long-acting risperidone was well tolerated locally and systematically. Although overall bioequivalence of the two formulations was established, the differences in pharmacokinetic profiles between the two formulations indicate potential benefits for long-acting risperidone.
Kemp, D. E., Canan, F., Goldstein, B. I., & McIntyre, R. S. (2009). Long-acting risperidone: a review of its role in the treatment of bipolar disorder. Advances in therapy, 26(6), 588–599. https://doi.org/10.1007/s12325-009-0039-0
Bipolar disorder is a multidimensional illness typified by fluctuating periods of depression and mania, cognitive dysfunction, abnormal circadian rhythms, and multiple comorbid psychiatric and general medical conditions. Indefinite pharmacological treatment is often required, yet the modest effects of available treatments and frequent difficulties with tolerability and adherence present complex challenges to patients. Long-acting injectable medications offer a therapeutic alternative to oral mood stabilizers and may help facilitate long-term treatment adherence. This article will provide a succinct review of the latest data on the use of long-acting injectable risperidone (LAR) during the maintenance phase treatment of bipolar disorder. The specific role of LAR in comparison to other atypical antipsychotics, and the limitations of available studies will be discussed from the perspectives of efficacy, tolerability, and sequential positioning in treatment guidelines.
Lasser, Robert A et al. “Clinical improvement in 336 stable chronically psychotic patients changed from oral to long-acting risperidone: a 12-month open trial.” The international journal of neuropsychopharmacology vol. 8,3 (2005): 427-38. doi:10.1017/S1461145705005225
Modern atypical antipsychotics have advantages over older neuroleptics. We hypothesize that their utility may be further enhanced by sustained drug delivery without daily oral self-dosing. This report examines the effects of a year of treatment with long-acting risperidone for chronically psychotic patients previously stabilized with oral risperidone. This open trial of long-acting risperidone involved 336 patients diagnosed with DSM-IV schizophrenia or schizoaffective disorder judged clinically stable on a consistent daily oral dose of risperidone for > or =4 wk. Based on oral doses, subjects were assigned clinically to bi-weekly intramuscular injections of 25-75 mg of long-acting risperidone for up to 50 wk. Clinical assessments at regular intervals included the Positive and Negative Syndrome Scale (PANSS), Clinical Global Impressions (CGI) scale, adverse event reports, and the Extrapyramidal Symptom Rating Scale (ESRS). PANSS total scores improved overall from a moderate baseline score of 64.5+/-17.7 to 58.8+/-19.9 at end-point (p<0.001), by> or =20% in 50% of patients, with greatest improvement in negative symptoms. Prevalence of favourable CGI - Severity ratings increased by 2.4-fold (p<0.0001). Ratings of extrapyramidal symptoms also improved [e.g. physician-rated parkinsonism scores decreased by 20% (p<0.0001)]. Tissue reactions and other adverse effects of repeated intramuscular injections were rare and mild. Psychotic patients considered stable but symptomatic with oral risperidone treatment showed further improvements in symptom ratings and extrapyramidal dysfunction during a year of bi-weekly injections of long-acting risperidone.
Additional documents
No documents were uploaded
Useful links
There are no additional links
Collaborate for development
Consider on a case by case basis, collaborating on developing long acting products with potential significant public health impact, especially for low- and middle-income countries (LMICs), utilising the referred to long-acting technology
Share technical information for match-making assessment
Provide necessary technical information to a potential partner, under confidentiality agreement, to enable preliminary assessment of whether specific medicines of public health importance in LMICs might be compatible with the referred to long-acting technology to achieve a public health benefit
Work with MPP to expand access in LMICs
In the event that a product using the referred to long-acting technology is successfully developed, the technology IP holder(s) will work with the Medicines Patent Pool towards putting in place the most appropriate strategy for timely and affordable access in low and middle-income countries, including through licensing